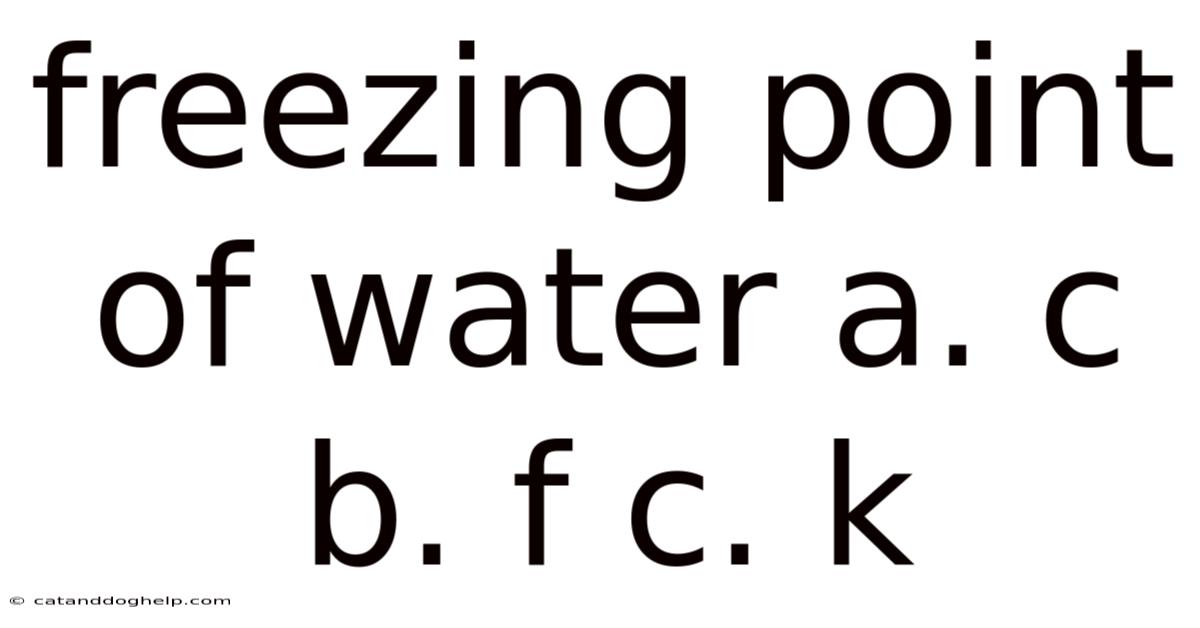Have you ever wondered why the roads get icy in winter or why your ice cream melts faster on a hot day? The secret lies in understanding the freezing point of water, a critical concept that impacts everything from weather patterns to cooking techniques. The freezing point isn't just a fixed number; it's a dynamic property influenced by various factors.
When we talk about freezing point, we're not just discussing the moment water turns to ice. We're diving into a world of molecular interactions, energy transfer, and the fundamental laws of physics. But whether you're a student learning about thermodynamics, a chef perfecting your frozen desserts, or simply curious about the world around you, grasping the concept of the freezing point of water is essential. Let's explore how this important point is measured across different scales: Celsius, Fahrenheit, and Kelvin Simple, but easy to overlook. And it works..
Main Subheading
Understanding the freezing point of water is more than just memorizing a number; it's about comprehending the behavior of molecules as they transition from a liquid to a solid state. Water, a ubiquitous substance, is key here in life and various scientific processes. Its freezing point is a key property that dictates many natural phenomena, from the formation of glaciers to the survival of aquatic life in cold climates.
The freezing point of a substance is defined as the temperature at which it changes from a liquid to a solid state. Also, for water, this transition occurs when the molecules lose enough kinetic energy to slow down and form stable hydrogen bonds, arranging themselves into a crystalline structure known as ice. In practice, this temperature is a specific point on various temperature scales, each with its own reference and interval. Whether you're using Celsius, Fahrenheit, or Kelvin, knowing the precise freezing point in each scale is vital for accurate scientific measurements and practical applications Most people skip this — try not to..
Comprehensive Overview
The freezing point of water is scientifically defined as the temperature at which the liquid and solid phases can coexist in equilibrium at a standard atmospheric pressure. This point is not arbitrary; it is determined by the physical properties of water molecules and their interactions. Water (H₂O) molecules are polar, meaning they have a slightly positive charge on the hydrogen atoms and a slightly negative charge on the oxygen atom. This polarity allows water molecules to form hydrogen bonds with each other, which are relatively strong intermolecular forces That's the part that actually makes a difference. And it works..
When water is cooled, the kinetic energy of its molecules decreases. So naturally, as the temperature drops, these molecules move more slowly, allowing hydrogen bonds to form more readily. At the freezing point, the kinetic energy is low enough that hydrogen bonds can stabilize into a crystalline lattice structure, resulting in the formation of ice. This process releases energy in the form of heat, known as the latent heat of fusion.
Historically, the establishment of the freezing point of water has been crucial for developing temperature scales. Anders Celsius, a Swedish astronomer, originally defined his scale in 1742 with 0 degrees as the boiling point of water and 100 degrees as the freezing point. Because of that, this was later inverted to the scale we use today. Similarly, Gabriel Fahrenheit used a mixture of ice, water, and salt to define 0 degrees on his scale, with the freezing point of water set at 32 degrees The details matter here..
The Kelvin scale, on the other hand, is an absolute temperature scale, meaning that its zero point is absolute zero – the theoretical point at which all molecular motion stops. The size of one Kelvin is the same as one degree Celsius, but the Kelvin scale starts at absolute zero (-273.That's why 15°C). That's why, the freezing point of water in Kelvin is 273.15 K. The Kelvin scale is widely used in scientific applications because it simplifies many thermodynamic calculations.
Understanding these scales and their relationship to the freezing point of water is essential in various fields. In meteorology, it helps predict weather patterns and understand climate changes. In chemistry and physics, it is used as a reference point for calibrating instruments and conducting experiments. In everyday life, it informs cooking, food storage, and even the design of infrastructure in cold climates. Knowing that water freezes at 0°C, 32°F, or 273.15 K provides a common ground for understanding and communicating temperature-related phenomena across different contexts.
Trends and Latest Developments
Current trends and research continue to refine our understanding of the freezing point of water under various conditions. As an example, the phenomenon of supercooling, where water remains in a liquid state below its freezing point, is an area of active research. While we commonly refer to the freezing point at standard atmospheric pressure, this point can shift under different pressures or when water contains impurities. Supercooling can occur in very pure water under specific conditions, and it has implications for cloud formation and atmospheric science.
This is the bit that actually matters in practice.
Another area of interest is the effect of dissolved substances on the freezing point. Still, adding salt or other solutes to water lowers its freezing point, a principle used in de-icing roads during winter. Recent studies are exploring the use of alternative de-icing agents that are less harmful to the environment but still effective in lowering the freezing point The details matter here..
Data from climate research also highlight the importance of the freezing point of water in understanding global warming. As temperatures rise, the melting of ice caps and glaciers is accelerating, contributing to sea-level rise and altering ocean salinity. Monitoring the freezing and thawing cycles of permafrost is crucial for predicting the release of greenhouse gases, such as methane, which can further exacerbate climate change Worth keeping that in mind. Turns out it matters..
Professional insights suggest that a comprehensive understanding of the freezing point of water is essential for addressing various environmental and technological challenges. Here's one way to look at it: advances in materials science are leading to the development of new types of ice-resistant coatings for aircraft and ships, which can prevent the formation of ice and improve safety. In the food industry, controlling the freezing process is critical for preserving food quality and extending shelf life.
On top of that, the study of water's freezing behavior at the nanoscale is opening new avenues in nanotechnology. So confined water in nanoscale spaces can exhibit drastically different freezing properties compared to bulk water, which has implications for designing new materials and devices. These developments underscore the ongoing relevance of the freezing point of water in both fundamental research and practical applications.
Tips and Expert Advice
Understanding the freezing point of water isn't just theoretical; it has practical applications in everyday life. Here are some tips and expert advice to help you apply this knowledge effectively:
-
Preventing Frozen Pipes: One of the most common issues during winter is frozen pipes. When water freezes, it expands, which can cause pipes to burst. To prevent this, insulate exposed pipes in unheated areas like basements and crawl spaces. During extremely cold weather, let faucets drip slightly to keep water moving through the pipes, reducing the risk of freezing.
-
De-icing Strategies: Knowing that salt lowers the freezing point of water can help you safely de-ice walkways and driveways. Rock salt (sodium chloride) is a common and effective option. Even so, be mindful of its potential impact on vegetation and concrete. Alternative de-icers, such as calcium chloride or magnesium chloride, are less harmful but may be more expensive. Always follow the manufacturer's instructions and use sparingly Most people skip this — try not to..
-
Optimizing Freezing for Food Preservation: Freezing is an excellent way to preserve food, but the rate of freezing can affect the quality. Rapid freezing helps to minimize the formation of large ice crystals, which can damage cell structures and result in a mushy texture when thawed. Use a blast chiller or freezer with a "quick freeze" setting for best results. Also, ensure food is properly packaged to prevent freezer burn, which occurs when moisture evaporates from the surface of the food.
-
Cooking and Baking: The freezing point of water plays a critical role in various culinary applications. Here's one way to look at it: when making ice cream, adding salt to the ice water surrounding the ice cream mixture lowers the freezing point, allowing the mixture to freeze faster and more evenly. In baking, understanding how the water content of ingredients affects dough consistency and freezing behavior is crucial for achieving the desired texture and rise.
-
Understanding Weather Patterns: Knowledge of the freezing point of water is essential for interpreting weather forecasts. When temperatures are near freezing, be aware of the potential for black ice – a thin, transparent layer of ice that forms on roads and sidewalks, making them extremely slippery. Also, understand the difference between freezing rain, sleet, and snow, all of which are related to the temperature profile of the atmosphere and the freezing point.
By applying these practical tips, you can use your understanding of the freezing point of water to improve safety, efficiency, and quality in various aspects of your daily life. Whether you're protecting your home from winter damage, preserving food, or interpreting weather conditions, this knowledge can make a significant difference.
FAQ
Q: What is the freezing point of pure water in Celsius? A: The freezing point of pure water is 0°C (zero degrees Celsius).
Q: What is the freezing point of water in Fahrenheit? A: The freezing point of water is 32°F (thirty-two degrees Fahrenheit).
Q: What is the freezing point of water in Kelvin? A: The freezing point of water is 273.15 K (two hundred seventy-three point fifteen Kelvin).
Q: Does pressure affect the freezing point of water? A: Yes, increasing pressure generally lowers the freezing point of water, although the effect is relatively small for moderate pressure changes.
Q: How does adding salt affect the freezing point of water? A: Adding salt lowers the freezing point of water, a phenomenon known as freezing point depression. This is why salt is used to de-ice roads.
Q: What is supercooling? A: Supercooling is the phenomenon where water remains in a liquid state below its freezing point without solidifying. This can occur in very pure water under specific conditions.
Q: Why is the freezing point of water important? A: The freezing point of water is important for various reasons, including understanding weather patterns, preserving food, preventing frozen pipes, and calibrating scientific instruments Small thing, real impact..
Conclusion
Simply put, the freezing point of water is a fundamental concept with far-reaching implications. Still, whether measured in Celsius (0°C), Fahrenheit (32°F), or Kelvin (273. Because of that, 15 K), this temperature marks the critical transition from liquid to solid, influencing everything from weather patterns to culinary techniques. Understanding the factors that affect the freezing point, such as pressure and impurities, allows for practical applications like preventing frozen pipes, de-icing roads, and optimizing food preservation.
By grasping the significance of the freezing point of water, we gain a deeper understanding of the world around us and can apply this knowledge to solve everyday challenges. Now that you're equipped with this information, we encourage you to explore further. Leave a comment sharing your own experiences with freezing temperatures or ask any questions you still have. Let's continue the conversation and expand our collective understanding of this fascinating phenomenon But it adds up..